Abstract
A 28-year-old Chinese man presented with acute bleeding per rectum. Computed tomography showed a posterior outpouching arising from the distal ileum. The outpouching had hyperaemic walls, but no active contrast extravasation was detected. Technetium-99m pertechnetate scintigraphy showed focal areas of abnormal uptake in the right side of the pelvis, superior and posterior to the urinary bladder. These areas of uptake appeared simultaneously with the gastric uptake and demonstrated gradual increase in intensity on subsequent images. The diagnosis of Meckel’s diverticulum was confirmed on surgery and the lesion was resected. The clinical and imaging features of Meckel’s diverticulum are discussed.
CASE PRESENTATION
A 28-year-old Chinese man presented to the accident and emergency department with active bleeding per rectum. The patient had experienced similar bouts of bleeding in the preceding two years and was treated symptomatically. He was stable on clinical examination and had normal blood pressure, pulse rate and respiratory rate. Although there was altered blood on digital rectal examination, no masses were felt. Computed tomography (CT) was performed to identify the possible cause of bleeding (
Fig. 1
Contrast-enhanced axial CT image.
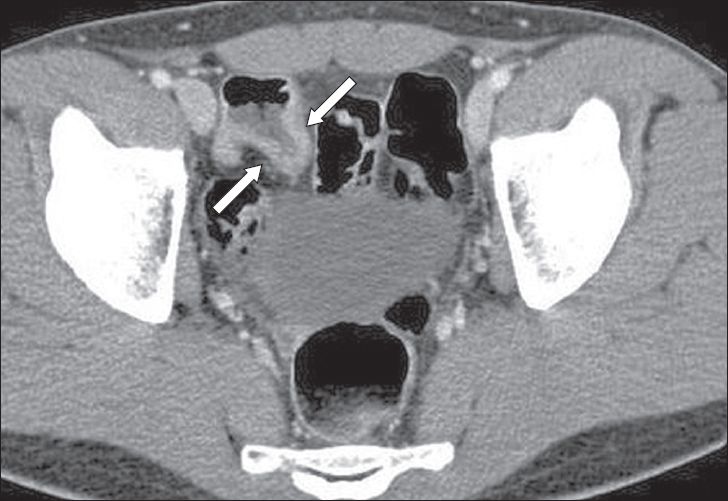
Fig. 2
Tc-99m pertechnetate (a) anterior planar scintigraphy and (b) sagittal single-photon emission computed tomography (SPECT)/CT fusion images.
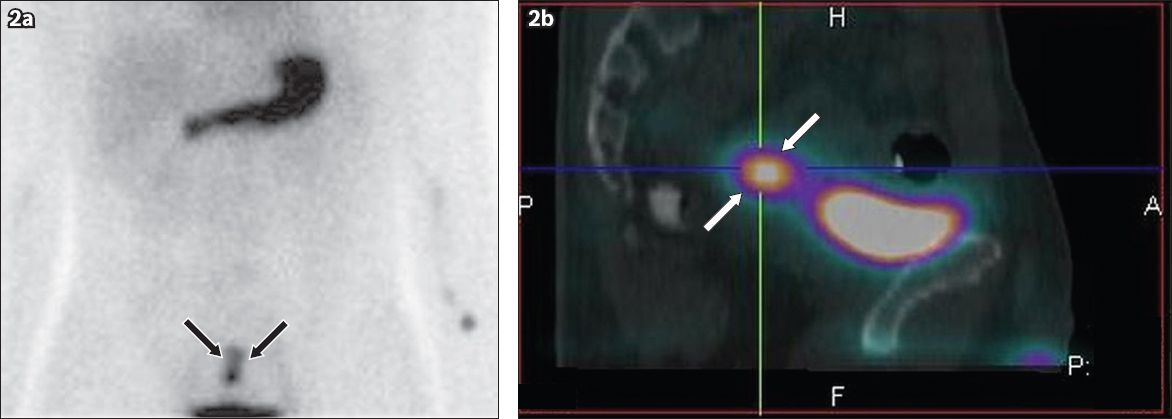
IMAGE INTERPRETATION
Contrast-enhanced axial CT image (
DIAGNOSIS
Meckel’s diverticulum.
CLINICAL COURSE
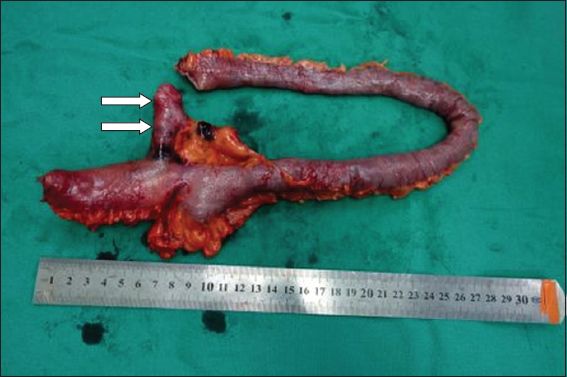
The imaging diagnosis was confirmed on retrograde single-balloon endoscopy, during which features of melanosis coli were seen. The distal ileum, from which biopsy samples were obtained, showed areas of scarring and multiple ulcerations. Subsequent laparoscopic evaluation confirmed the presence of the Meckel’s diverticulum, which arose about 62 cm from the ileocaecal junction. Small bowel resection was performed to incorporate the diverticulum and the areas of ulceration. The resected bowel, which measured 28 cm in length and included a 4.5-cm long outpouching from the antimesenteric surface (
Fig. 3
Photograph of the resected specimen shows a short length of ileum with posterior outpouching (arrows), consistent with a Meckel’s diverticulum.
DISCUSSION
Meckel’s diverticulum is the most common congenital anomaly of the gastrointestinal tract and is found in about 2% of the general population.(1,2) It was first described in 1809 by Johann Friedrich Meckel and is slightly more common in men.(2) It contains all three layers of the intestinal wall and hence is a true diverticulum. Originating from the antimesenteric border of the distal ileum, it arises due to the incomplete closure of the omphalomesenteric duct. In the majority of cases, this diverticulum is located about 100 cm from the ileocaecal valve. This diverticulum can vary in length and width, to as long as 10 cm in some instances. Meckel’s diverticulum can contain heterotopic mucosa, commonly the gastric or pancreatic mucosa.(1,2) Heterotopic gastric mucosa is more common than duodenal or jejunal mucosa and can result in ulcerations, which can subsequently cause gastrointestinal bleeding. Vascular supply to the diverticulum is mostly from the omphalomesenteric artery, which arises from one of the ileal branches of the superior mesenteric artery.
Meckel’s diverticulum usually remains asymptomatic throughout life; however, it can also present with a number of well-known complications. These complications may range from varying degrees of inflammation, gastrointestinal tract bleeding(3-5) and small bowel obstruction(6) to perforation. Patients often present with acute abdominal pain localised to the right lower quadrant. The estimated lifetime risk of complications in patients with this diverticulum is 4%.(1) Gastrointestinal haemorrhage is a serious presentation that is more common and severe in children.(7) The bleeding is due to heterotopic gastric mucosa, which results in ulcerations also seen in the adjacent loops of ileum. Another common complication is acute diverticulitis, which may be of varying degrees of severity. The orifice of the diverticulum may be blocked by an enterolith or retained foreign bodies,(8) resulting in stasis and infection. The diverticulum may also undergo torsion, resulting in inflammation or ischaemia. Acute inflammation of a Meckel’s diverticulum is a well-known mimic of acute appendicitis. Severe cases of inflammation may result in perforation, which is a life-threatening complication. Many other conditions involving the lower abdomen and pelvis can also present with complaints similar to those of Meckel’s diverticulum, thus making Meckel’s diverticulum extremely difficult to diagnose based on clinical features alone. Small bowel obstruction is also a well-known complication and can occur due to the twisting of the bowel at the site of the diverticulum. Larger diverticula have greater chances of causing obstruction and may also be associated with intussusceptions.(9) Although rarely seen, a large Meckel’s diverticulum may invert within the distal ileum and cause obstruction. Carcinoid tumours arising within Meckel’s diverticula are rare,(10) although they are the most commonly associated neoplasm. Malignant neoplasms arising from the diverticulum are extremely rare.
Imaging plays an extremely important role in evaluating patients with suspected Meckel’s diverticulum. Radiographs of the abdomen are usually unremarkable unless there are features of intestinal obstruction or perforation. Ultrasonography is often performed in children as the first imaging modality. However, the diagnosis is extremely difficult; Meckel’s diverticulum is similar in appearance to the appendix, although it is in a different location. The diverticulum is visualised as a blind-ending, tubular echogenic structure that has clear communication with a small bowel loop. Contrast-enhanced CT of the abdomen plays a very important role in diagnosis and can also identify associated complications in the majority of cases. The diverticulum is visualised as a blind-ending tubular structure arising from the antimesenteric border of the ileum, a short distance proximal to the ileocaecal junction (
Fig. 4
A 35-year-old woman was evaluated for left iliac fossa pain. Contrast-enhanced coronal CT image shows a large blind-ending tubular structure arising from the small bowel (arrows). This incidental finding is a Meckel’s diverticulum.
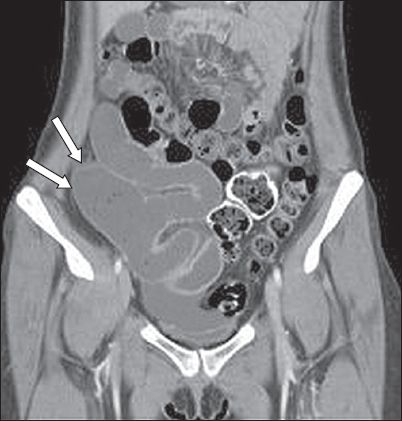
Fig. 5
A 25-year-old man presented with colicky right iliac fossa pain of one-day duration. (a) Coronal CT scout image shows multiple grossly dilated small bowel loops. (b) Contrast-enhanced coronal CT image shows faecal-laden distal ileum and a Meckel’s diverticulum (arrows).
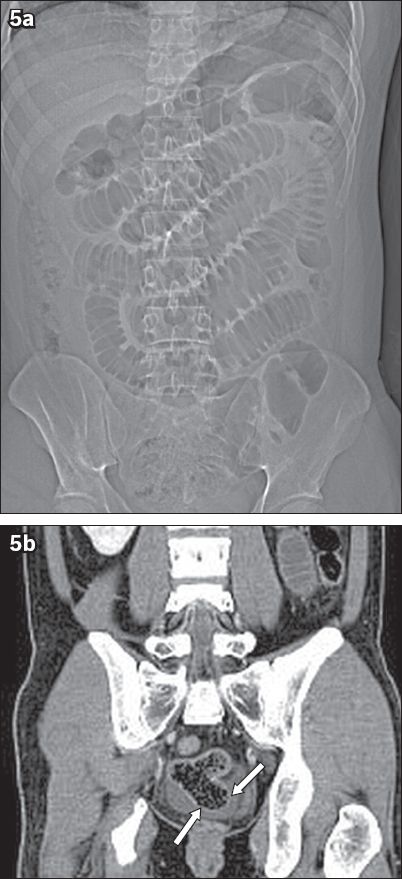
Fig. 6
A 40-year-old Chinese man presented with acute onset severe lower abdominal pain. Contrast-enhanced coronal CT image shows an inflamed Meckel’s diverticulum (arrows) with surrounding free fluid and fat stranding (arrowheads), typical of diverticulitis.
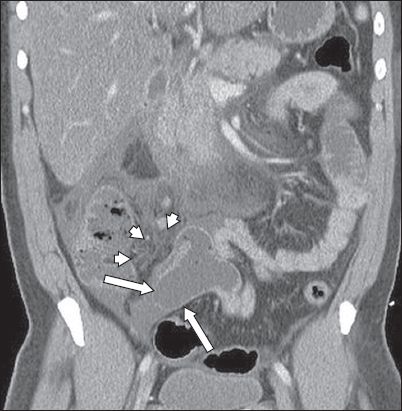
Fig. 7
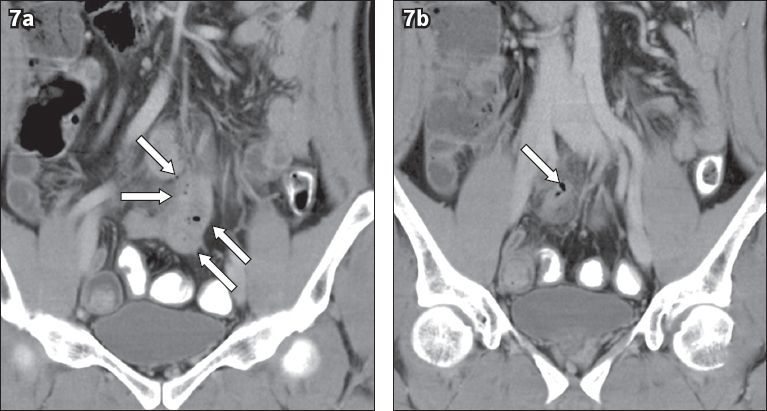
A 49-year-old man complained of acute onset epigastric discomfort. (a) Contrast-enhanced coronal CT image shows an outpouching from the distal ileum (arrows) with pockets of free air in the small bowel mesentery (arrowheads), consistent with perforation. (b) Contrast-enhanced coronal CT image shows the inflamed Meckel’s diverticulum (arrow).
Nuclear medicine scintigraphy also plays an important role in identifying Meckel’s diverticulum; however, its role is limited to cases with heterotopic gastric mucosa.(11) Overall diagnostic accuracy in these cases may be up to 90%.(1) The overall sensitivity of Tc-99m pertechnetate scintigraphy is about 60% in cases of Meckel’s diverticulum. The uptake of pertechnetate by the ectopic gastric mucosa in the diverticulum is focal and simultaneous with the uptake of pertechnetate by the stomach, and gradually increases on subsequent phases of imaging. It is important to correlate the scintigraphic findings with CT imaging, as many of these cases are a diagnostic challenge.
Timely diagnosis of a complicated Meckel’s diverticulum is very important, as the management varies depending on the severity of the complications. Surgery is usually indicated in cases of symptomatic diverticula, especially those presenting with bleeding. The decision for surgery also depends on the overall condition of the patient. During surgery, it is important to resect all foci of the ectopic mucosa and look for associated ulcerations, which are typically seen at the junction of the normal and ectopic mucosa. Asymptomatic patients are conservatively managed and surgery is usually not performed. Meckel’s diverticula can also be incidentally detected during laparotomy and their treatment is controversial. The choice of resection in such cases depends on the surgeon, overall condition of the patient and extent of the surgery being performed.
In summary, Meckel’s diverticulum is a well-known cause of right lower quadrant pain, especially in young patients. The clinical presentation can often overlap with a number of other pathologies, making the diagnosis difficult. Contrast-enhanced CT, in conjunction with Tc-99m pertechnetate scintigraphy, can aid radiologists in making a confident diagnosis. Symptomatic patients usually require early surgery to resect the diverticulum.


